

Products
This material is certified for
FDA – 21 CFR
& is compliant with
USP CLASS VI & ISO 10993
CRYOMATRIX
VITRIFICATION STRAW VIDEO
Vitrification is a technology followed worldwide for freezing of eggs and embryos cryopreservation as well. Cryomatrix is open carrier device which gels well into any media and is a comfortable partner in vitrification process of Human or Animal IVF labs and in research centers.
Cryomatrix is made from medical grade polystyrene materials which meets ISO 1133/ISO 527/ISO 306 & IEC – 60695-11-10, for its physical, mechanical and thermal properties. This material is certified for FDA – 21 CFR & is compliant with USP CLASS VI and ISO 10993.

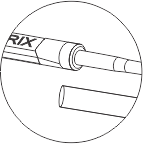
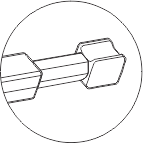
CRYOMATRIX Usage & Design


CRYOMATRIX Features
- It is manufactured in complete clean room - ensuring product is free from Human DNA, RNA free, DNA free and Endotoxin free.
- It is manufactured under quality ISO 13485:2016 standard.
- It is individually packed gamma radiation sterilized to ensure sterility of the product.
- It has a curved shaped tip to enhance safe and easy loading/ unloading of specimen.
- It is designed in such a way that it has no buoyancy effect and sinks in LN easily.
- It is subjected to MEA and Endotoxin study -- batch to batch in well-known certified lab in USA.
- It has been designed to provide catchment area for forceps for smooth locking and unlocking of device.
- The curved shaped tip ensures the specimens are safe during locking and unlocking of the device. It is available is 6 different trans lucid colors: pack of 10 units per box.
Certificates
Kindly write us an email at info@cryobiomatrix.com.
Please mention the lot number and we will send you the certificate of analysis.
Distributors